
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Search
- Page Path
- HOME > Search
Response
- Altered Metabolic Phenotypes and Hypothalamic Neuronal Activity Triggered by Sodium-Glucose Cotransporter 2 Inhibition (Diabetes Metab J 2023;47:784-95)
- Ho Gyun Lee, Il Hyeon Jung, Byong Seo Park, Hye Rim Yang, Kwang Kon Kim, Thai Hien Tu, Jung-Yong Yeh, Sewon Lee, Sunggu Yang, Byung Ju Lee, Jae Geun Kim, Il Seong Nam-Goong
- Diabetes Metab J. 2024;48(1):159-160. Published online January 29, 2024
- DOI: https://doi.org/10.4093/dmj.2022.0458
- 712 View
- 93 Download
Original Articles
- Basic Research
- Altered Metabolic Phenotypes and Hypothalamic Neuronal Activity Triggered by Sodium-Glucose Cotransporter 2 Inhibition
- Ho Gyun Lee, Il Hyeon Jung, Byong Seo Park, Hye Rim Yang, Kwang Kon Kim, Thai Hien Tu, Jung-Yong Yeh, Sewon Lee, Sunggu Yang, Byung Ju Lee, Jae Geun Kim, Il Seong Nam-Goong
- Diabetes Metab J. 2023;47(6):784-795. Published online August 23, 2023
- DOI: https://doi.org/10.4093/dmj.2022.0261
- 1,506 View
- 149 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
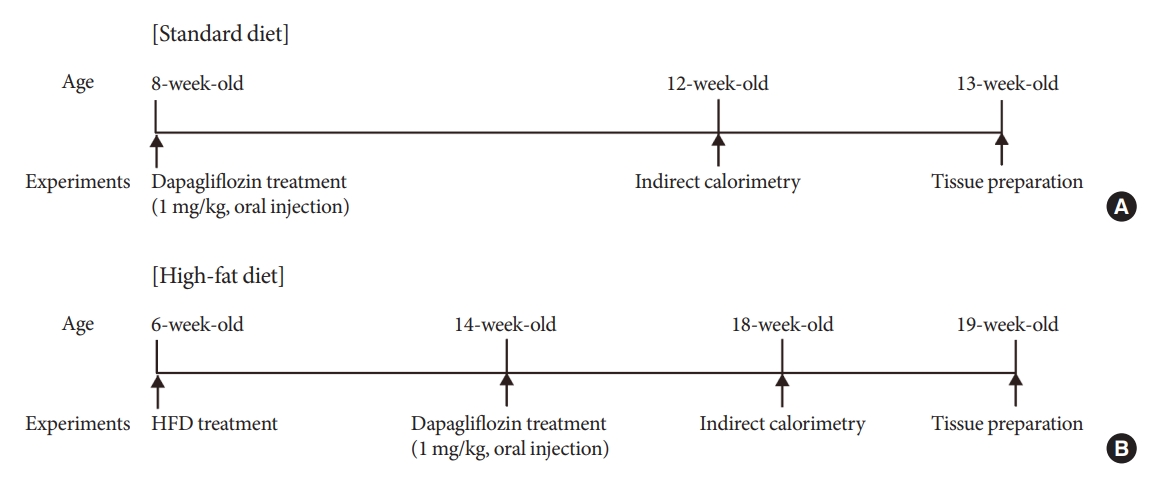
Sodium-glucose cotransporter 2 (SGLT-2) inhibitors are currently used to treat patients with diabetes. Previous studies have demonstrated that treatment with SGLT-2 inhibitors is accompanied by altered metabolic phenotypes. However, it has not been investigated whether the hypothalamic circuit participates in the development of the compensatory metabolic phenotypes triggered by the treatment with SGLT-2 inhibitors.
Methods
Mice were fed a standard diet or high-fat diet and treated with dapagliflozin, an SGLT-2 inhibitor. Food intake and energy expenditure were observed using indirect calorimetry system. The activity of hypothalamic neurons in response to dapagliflozin treatment was evaluated by immunohistochemistry with c-Fos antibody. Quantitative real-time polymerase chain reaction was performed to determine gene expression patterns in the hypothalamus of dapagliflozin-treated mice.
Results
Dapagliflozin-treated mice displayed enhanced food intake and reduced energy expenditure. Altered neuronal activities were observed in multiple hypothalamic nuclei in association with appetite regulation. Additionally, we found elevated immunosignals of agouti-related peptide neurons in the paraventricular nucleus of the hypothalamus.
Conclusion
This study suggests the functional involvement of the hypothalamus in the development of the compensatory metabolic phenotypes induced by SGLT-2 inhibitor treatment. -
Citations
Citations to this article as recorded by- Altered Metabolic Phenotypes and Hypothalamic Neuronal Activity Triggered by Sodium-Glucose Cotransporter 2 Inhibition (Diabetes Metab J 2023;47:784-95)
Jae Hyun Bae
Diabetes & Metabolism Journal.2024; 48(1): 157. CrossRef - Altered Metabolic Phenotypes and Hypothalamic Neuronal Activity Triggered by Sodium-Glucose Cotransporter 2 Inhibition (Diabetes Metab J 2023;47:784-95)
Ho Gyun Lee, Il Hyeon Jung, Byong Seo Park, Hye Rim Yang, Kwang Kon Kim, Thai Hien Tu, Jung-Yong Yeh, Sewon Lee, Sunggu Yang, Byung Ju Lee, Jae Geun Kim, Il Seong Nam-Goong
Diabetes & Metabolism Journal.2024; 48(1): 159. CrossRef
- Altered Metabolic Phenotypes and Hypothalamic Neuronal Activity Triggered by Sodium-Glucose Cotransporter 2 Inhibition (Diabetes Metab J 2023;47:784-95)
- The Role of Hypothalamic FoxO1 on Hyperphagia in Streptozotocin-Induced Diabetic Mice.
- Il Seong Nam-Goong, Jae Geun Kim, Se Jin Kim, Seong Jae Hur, Jin Woo Lee, Eun Sook Kim, Chang Ho Yun, Byung Ju Lee, Young Il Kim
- Korean Diabetes J. 2009;33(5):375-381. Published online October 1, 2009
- DOI: https://doi.org/10.4093/kdj.2009.33.5.375
- 1,962 View
- 22 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Streptozotocin-induced diabetic animals are characterized by hyperphagia due to deficiencies of insulin and leptin. Forkhead box-containing protein of the O subfamily-1 (FoxO1) regulates energy homeostasis by regulating energy expenditure and food intake as well as mediating insulin and leptin signals in the hypothalamus. To identify the mediator of diabetic hyperphagia, we examined the effects of insulin or leptin on hypothalamic FoxO1 expression in a diabetic animal model. METHODS: Diabetes was induced in mice (C57BL/6) by intraperitoneal administration of streptozotocin (200 mg/kg). Stainless steel cannula was implanted into the lateral ventricle of the brain in each mouse. After three weeks, the mice were administered saline, insulin or leptin via intracerebroventricular (ICV) route. The medial hypothalamus was isolated to evaluate the mRNA expressions of FoxO1 and neuropeptides. RESULTS: Streptozotocin-induced diabetic mice exhibited significant elevations of blood glucose and food intake and significantly low levels of serum insulin and leptin. The levels of hypothalamic FoxO1 mRNA were significantly increased in diabetic mice. The hypothalamic expression of neuropeptide Y (NPY) mRNA was increased, but the expression of preproopiomelanocortin (POMC) mRNA was decreased in diabetic mice. ICV administration of insulin or leptin attenuated the upregulation of hypothalamic FoxO1 mRNA, and resulted in downregulation of NPY mRNA and upregulation of POMC mRNA in diabetic mice. CONCLUSION: We observed that the expression of hypothalamic FoxO1 mRNA was increased in streptozotocin-induced diabetic mice, and that it was significantly attenuated by central administration of insulin or leptin. These results suggest that hypothalamic FoxO1 is the direct mediator of diabetic hyperphagia.

 KDA
KDA
 First
First Prev
Prev





